Current Projects
NEW AWARD! 1R61AG090384 Off-the-shelf CAR-Engineered Macrophage Therapy for Alzheimer’s Disease
Aβ plaques and hyperphosphorylated tau tangles are two main pathological hallmarks of Alzheimer’s disease. They also are considered the primary targets for developing disease modifying therapies. Very recently, monoclonal antibodies (mAbs) targeting Aβ plaques have been approved by the US Federal Drug Administration for the treatment of Early Alzheimer’s Disease. This novel therapy has been proven effective in reversing Aβ plaque deposition and slowing down the rate of cognitive decline in Alzheimer patients, yet its use comes with several important limitations. These include high cost, a requirement for continuous mAb infusions, and frequent adverse effects in form of Amyloid Related Imaging Abnormalities, which include focal brain edema and microhemorrhages. Occurrence of these adverse effects often requires pausing of the treatment or even its permanent discontinuation, which limits its efficacy.
In recent years, Chimeric Antigen Receptor (CAR) cell therapies have established themselves as a breakthrough treatments for several types of blood cancer. A CAR is an engineered receptor protein which can be expressed in immune cells—typically T lymphocytes (CAR-T) or macrophages (CAR-M)—and makes them capable of targeted destruction of cancer cells.
We have proposed the development of modified macrophages expressing CARs targeting Aβ: CAR-Aβ-iMφ therapy for Alzheimer’s disease. CAR-Aβ-iMφ cells will be engineered to penetrate the brain and remove Aβ plaques eliminating the need for continuous administration of anti-Aβ mAbs and possibility of adverse effects associated with their use. Our CAR-Aβ-iMφ cells also will be designed to attenuate neuroinflammation, which is associated with Alzheimer disease pathology and substantially contributes to its progression.
Role of Astrocytes in the Alzheimer Pathobiology
Astrocytes are the most numerous brain cells, and their primary function is to support complex metabolic needs of neurons. In Alzheimer’s disease, and in other neurodegenerative diseases, astrocytes transition to a reactive, neurodegenerative state, where they display strong proinflammatory characteristics and become deleterious to already diseased neurons. The transition of astrocytes to the reactive state is induced by the signaling from neurodegenerative microglia. Communication between reactive astrocytes and neurodegenerative microglia in Alzheimer’s disease is bidirectional. Reactive astrocytes reciprocally stimulate neurodegenerative microglia and this detrimental crosstalk between both types of cells effectively drives the inflammatory cascade, which together with the b-amyloid deposition and the accumulation of hyperphosphorylated tau form three essential mechanisms of Alzheimer pathogenesis. In this exploratory project we aim to identify and characterize genetic factors endowing astrocytes with resistance to the transformation into reactive neurodegenerative state. Overexpression of these factors can be leveraged to attenuate neuroinflammation and thus used as a specific therapy for halting progression of Alzheimer’s disease.
This project is funded by the Fisher Center for Alzheimer’s Research Foundation
R01AG075840 Role of Microglia in Neurodegeneration – Effect of ApoE
In many neurodegenerative disorders including Alzheimer’s and prion diseases, an accumulation of misfolded proteins is exacerbated by a neurotoxic presentation of microglia, referred to as microglia neurodegenerative phenotype (MGnD). In addition to pro-inflammatory characteristics and a loss of homeostatic function, these neurotoxic microglia also express lipid-poor apolipoprotein E (apoE). Astrocytic release of lipid-rich apoE is thought to be protective by facilitating clearance of misfolded proteins, but microglial expression of apoE promotes their harmful pro-inflammatory features.
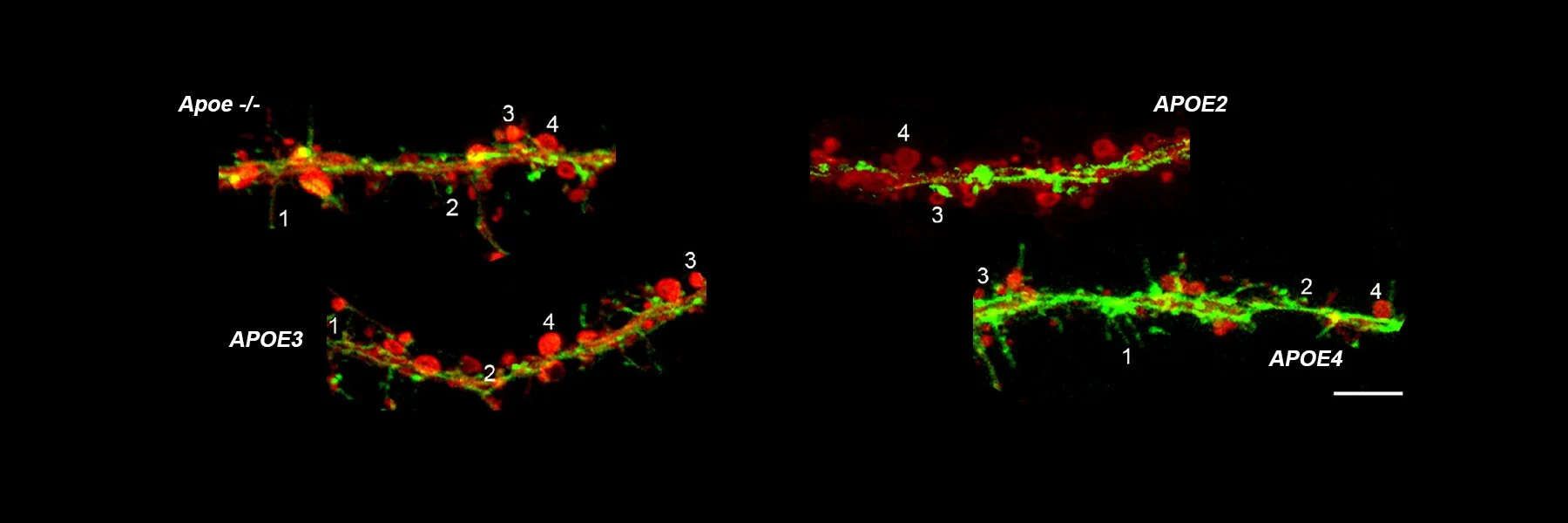
ApoE is encoded by the gene APOE, for which humans have three alleles—APOE ε2, ε3, and ε4, the last of which is well-known to vastly increase the risk of sporadic Alzheimer’s disease. One possible way to account for this increased risk is the isoform-specific nature of apoE which may relate to impaired clearance of Aβ in the case of APOE ε4 carriers. However, what remains to be understood are the specific effects of apoE lipidation, APOE genotype-driven differences in apoE, and astrocyte-derived vs. microglia-derived apoE on neurodegeneration.
To examine these mechanisms, scrapie-prion (PrP-Sc) inoculation will induce a state of neurodegeneration in mice, and effects will be explored of globally or conditionally knocking out ApoE from each cell source and preventing or increasing apoE lipidation. We will also characterize isolated microglia and astrocytes from each genetic background (APOE ε2, ε3, and ε4). This investigation will not only unlock important key elements in the mechanisms underlying apoE’s influence on neurodegenerative disorders, but it will also provide invaluable insights for the most vulnerable populations.
RF1AG088226 Apolipoprotein E genotype modulates brain mitovesicle production, a component of mitochondrial quality control
APOE ε4 allele carriers are not only at greater risk for sporadic Alzheimer’s disease, but they also tend to display quicker pathological progression. While it is understood that the APOE allele encodes apolipoprotein E (apoE) that may be helpful (ε2) or deleterious (ε4) to neuronal health in a genotype-dependent manner, the molecular and cellular underpinnings of this differential apoE effect represents a major knowledge gap. The specific role played by apoE lipoparticles in influencing the local regulation of nearby neurons has not yet been made clear.
Though mice do not have APOE allele diversity, we are examining neuronal morphology of primary neuronal cultures treated with apoE lipoparticles from transgenic mice with human APOE ε2, APOE ε3, or APOE ε4. These cultures will subsequently be analyzed for quantity and quality mitovesicles.
Mitovesicles are a newly discovered extracellular vesicle believed to be important for mitochrondrial homeostasis and thought to contribute to synaptotoxicity when excreted by diseased neurons. Mitochondria are the primary organelle responsible for energy generation in cells including neurons and become dysfunctional during both healthy aging and in neurodegenerative disorders. Dr. Efrat Levy’s lab has demonstrated that the amount and content of these mitovesicles are impacted by mitochondrial dysfunction associated with Alzheimer’s disease. This project will examine the hypothesis that mitovesicle production is a protective feature of mitochondrial quality control, improved by APOE ε2 and hindered by APOE ε4, and will establish imperative foundations for approaching clinical care for APOE ε4 carriers.
R01AG067478 Mechanisms of Peroxiredoxin 6 Endowed Protection in Alzheimer’s Disease
Beta-amyloid (Aβ) accumulation in the brain is the primary feature of preclinical Alzheimer’s disease, and the rate of deposition coincides with neurofibrillary degeneration and cognitive decline. Glial cells in central nervous system are integral to maintaining homeostasis in the brain and are therefore critically important in clearing the buildup of Aβ. Microglia and astrocytes both become activated and engage amyloid plaques in the brain for Aβ proteostasis during early Alzheimer’s disease.
Second to the APOE ε4 allele, Alzheimer’s-associated genetic variants in the TREM2 receptor in microglia have been identified as a major risk factor for sporadic Alzheimer’s disease. These TREM2 receptor variants render microglia inept at clearing Aβ and are largely responsible for ensuing pathological progression. Unlike microglia, the molecular features of astrocytes that govern their positive or negative contributions to Alzheimer’s pathology have not been elucidated.
Our preliminary experiments have led us to hypothesize that peroxiredoxin (Prdx) 6, a protein exclusively expressed by astrocytes in the central nervous system, may regulate peri-plaque activation of astrocytes and may importantly control crosstalk known to occur between astrocytes and microglia in their homeostatic efforts. Overexpressing Prdx6 seems to endow astrocytes and microglia with an enhanced protective effect via astrocytic and microglial enticement to amyloid plaques, making this an appealing therapeutic target for Alzheimer’s disease.
For more published research, visit our publications page